Revolutionizing Drug Discovery with Advanced Molecular Microscopy
The landscape of drug discovery is undergoing a significant transformation as advancements in molecular microscopy emerge. Central to this revolution are high-resolution imaging techniques, such as 3D microscopy and cryo-electron microscopy (cryo-EM), that allow researchers to visualize cellular processes with unprecedented clarity. This article addresses a fundamental question: How are these advanced microscopy techniques reshaping the drug discovery process? This matter is crucial as traditional drug discovery methodologies often fall short in efficiently identifying viable drug candidates and predicting toxicities. A common misconception in this field is that high-throughput screening alone is adequate for drug discovery; however, nuanced insights into cellular behavior and molecular interactions are essential for the development of effective therapies. Readers will learn how tools like the ZEISS LSM 910 confocal microscope and cryo-EM are powering a new era in drug development by enhancing our understanding of human biology at a molecular level.
Understanding Molecular Microscopy Techniques
Overview of Advanced Microscopy
At the forefront of this transformation are two pivotal technologies: high-resolution 3D imaging and cryo-electron microscopy (cryo-EM). Each technique has unique capabilities that contribute to drug discovery.
3D Microscopy: The ZEISS LSM 910 Confocal Microscope
The ZEISS LSM 910 confocal microscope utilizes Airyscan technology to achieve deep optical sectioning. This capability is vital for creating detailed 3D maps that reveal intricate spatial contexts of cells and their responses to drugs. Key features include:
- High-resolution imaging, enabling observation of subcellular details.
- Precise quantification of cellular behavior and gene expression.
- Enhanced visualization for reliable identification of drug candidates.
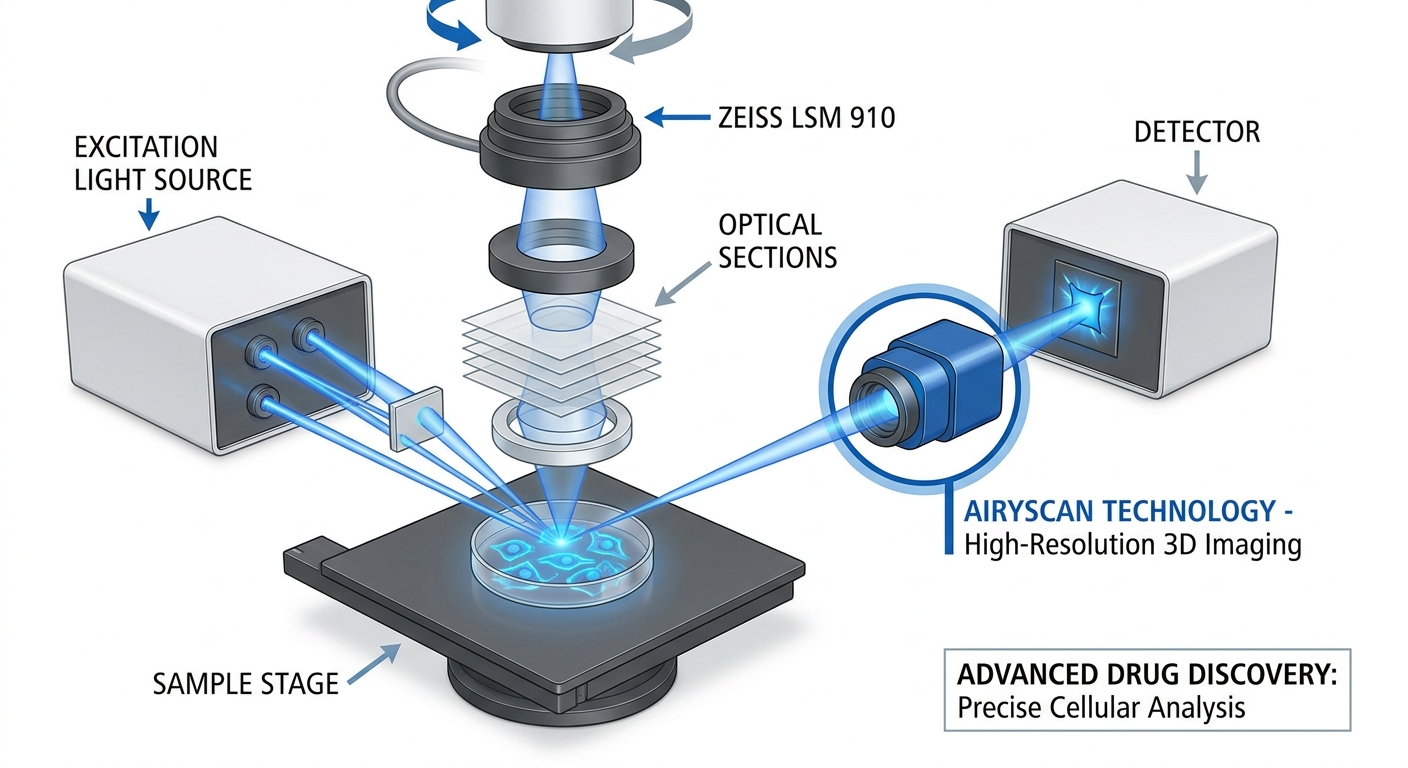
This advanced imaging facilitates early toxicology predictions, significantly accelerating the development of personalized therapies. For example, studies using organ-on-a-chip models analyzed via the ZEISS LSM 910 have demonstrated how drugs interact within a more accurate biological context, thereby fostering a deeper understanding of cellular responses.
Cryo-Electron Microscopy (Cryo-EM)
Cryo-EM has matured rapidly, emerging as a powerful tool for structural biology. This technique allows researchers to visualize molecular structures at atomic resolution, thereby revolutionizing rational drug design. Key benefits of cryo-EM include:
- Rapid epitope mapping for antibody therapeutics and immune response profiling.
- Insights into mechanisms of action for various therapeutic targets.
- Reduced time and costs associated with clinical development through early protein structure determination.
Notably, cryo-EM has enabled significant advancements in vaccine design. The ability to characterize viral particles and improve antigen identification can shorten the development cycles necessary to combat infectious diseases.
Comparative Benefits of High-Resolution Microscopy Techniques
To fully appreciate the impact of these advanced microscopy methods on drug discovery, a comparison of their capabilities is beneficial.
| Feature | ZEISS LSM 910 Confocal Microscope | Cryo-Electron Microscopy |
|---|---|---|
| Imaging Depth | Deep optical sectioning | Atomic-scale resolution |
| Cellular Context | 3D mapping of cellular responses | Protein structure determination |
| Applications | Drug candidate identification, toxicity prediction | Rational drug design, antibody discovery |
This comparison showcases the strengths of each methodology in enhancing drug discovery pipelines.
Impact on Drug Discovery Pipelines
Enhanced Drug Candidate Identification
Both microscopy techniques facilitate a more informed approach to candidate drug identification. For instance, the ZEISS LSM 910’s imaging capabilities allow for a thorough visualization of drug interactions at the cellular level. This rich dataset enables researchers to make more confident decisions about a candidate’s potential efficacy and safety profiles.

Predicting Toxicities Early
Effective drug development is heavily reliant on a robust understanding of a compound’s pharmacodynamics and pharmacokinetics. Early toxicity predictions can save both time and resources. The nuanced insights gained from 3D modeling and advanced imaging of cellular systems provide critical information about potential adverse effects, allowing researchers to modify compounds before clinical trials.
Personalized Medicine and Accelerated Development
With the insights gained from these advanced microscopy technologies, drug development can be tailored to individual patient profiles. This aligns with the shift towards personalized medicine, where treatments are increasingly customized based on a patient’s unique biological makeup. The ability to accelerate this process is vital, especially in response to emerging health crises.
Future Directions in Drug Discovery
The integration of advanced microscopy techniques such as the ZEISS LSM 910 and cryo-EM signifies a paradigm shift in drug discovery. As these technologies continue to evolve, we can expect:
- Higher resolution images, leading to improved understanding of cellular and molecular interactions.
- Enhanced automation in imaging processes, thereby expanding accessibility for researchers.
- Greater collaboration between pharmaceutical companies and technology developers to innovate methods that streamline drug discovery workflows.
Ultimately, these advancements hold the promise of not only accelerating drug development timelines but also refining the precision of therapeutic targeting.
FAQ
Q: What is the primary advantage of using 3D microscopy in drug discovery?
A: The primary advantage is the ability to visualize nuanced cellular behavior and responses in a three-dimensional context, which aids in more accurate drug candidate identification and toxicity predictions.
Q: How does cryo-EM contribute to rational drug design?
A: Cryo-EM allows for high-resolution imaging of molecular structures, enabling researchers to understand target interactions at an atomic level, which is crucial for designing effective therapeutics.
Q: Can advanced microscopy techniques reduce the cost of drug development?
A: Yes, by providing early insights into drug efficacy and toxicity, these techniques can minimize the risks and costs associated with late-stage clinical failures.
Q: Are these technologies applicable only to new drug development?
A: No, these technologies can also enhance the understanding of existing drugs and may lead to new indications or improved formulations.
Q: What is the future of microscopy in drug discovery?
A: The future includes further enhancements in imaging resolution, greater accessibility to researchers, and increased integration with computational models to predict drug responses effectively.
Conclusion
The advancements in molecular microscopy technologies such as the ZEISS LSM 910 and cryo-Electron microscopy are redefining drug discovery pipelines. Their capabilities enhance the identification of drug candidates, predict toxicities, and contribute to the evolution of personalized medicine. Future developments in microscopy are likely to drive even greater innovations in the field, making significant contributions to our understanding of complex biological systems and therapeutic needs.
For more on the foundational principles of microscopy, see Wikipedia: Microscopy.
Rotating USB



